产品详情
- ProductSizeAmountActiveMax® Human CD19 μBeads, premium grade (for cells)2.5 mg2.5 × 10⁷ beadsActiveMax® Human CD19 μBeads, premium grade (for cells)10 mg (2.5 mg × 4)1.0 × 10⁸ beads
背景(Background)
ActiveMax® Human CD19 μBeads, premium grade (for cells) are produced under sterile manufacturing conditions (ISO 5), and no animal- or human-derived components are used throughout the production process. It is produced under our rigorous quality control system that includes a comprehensive set of tests including sterility and endotoxin tests.
表达区间及表达系统(Source)
ActiveMax® Human CD19 μBeads, premium grade (for cells) are uniform, superparamagnetic beads of 5.5 µm with streptavidin on its surface and coupled with biotinylated Human CD19 Protein, expressed from human 293 cells (HEK293) and contains AA Pro 20 - Lys 291 (Accession # P15391-1).
应用说明(Application)
ActiveMax® Human CD19 μBeads, premium grade (for cells) are designed to stimulate in vitro CD19-specific CAR-T cells or UCAR-T cells, similar to the tumor cell lines that express human CD19 antigen. It can be used as follows:
Evaluating the characteristics of CAR-T cells or UCAR-T cells.
In vitro expansion of CD19-specific CAR-T cells or UCAR-T cells.
In vitro enrichment of CD19-specific CAR-T cells or UCAR-T cells.* We also carry ActiveMax® Streptavidin μBeads, premium grade (for cells)(Cat. No. MBS-C009), and it can be used for convenient preparation of your own sterile protein/antibody coupled beads for cell-based assay, cell sorting, and other applications. The ActiveMax® Streptavidin μBeads, premium grade can also be used together with other ActiveMax® μBeads, premium grade as Isotype control.
重构方法(Reconstitution)
See Certificate of Analysis (CoA) for detailed instruction.
存储(Storage)
This product is stable in storage under the following conditions: -20˚C for 12 months in lyophilized state. -70°C for 3 months under sterile conditions after reconstitution.
Please avoid repeated freeze-thaw cycles after reconstitution. Immediate use after reconstitution is highly recommended.
无菌(Sterility)
Negative
内毒素(Endotoxin)
Less than 0.002 EU per μg by the LAL method.
注意事项(Important Note)
This product is for research use only and not intended for therapeutic or in vivo diagnostic use.
制剂(Formulation)
Please contact us for detailed information.
Contact us for customized product form or formulation.
质量管理控制体系(QMS)
产品展示
典型数据-Typical Data
Please refer to DS document for the assay protocol.
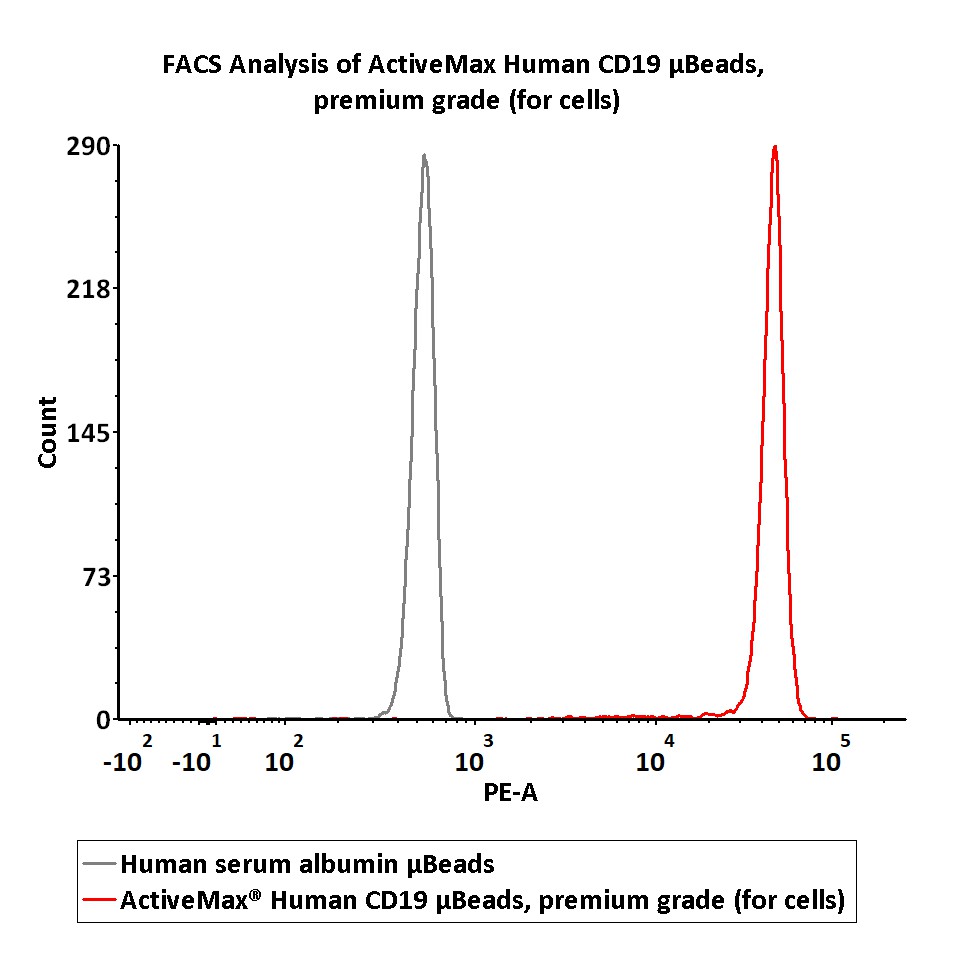
Assay of human CD19 protein on the μBeads surface by Flow cytomtry. The human CD19 conjugated on the μBeads (Cat. No. MBS-C002) surface were fluorescently stained using PE labeled anti-human CD19 antibody and analyzed by flow cytometry (QC tested).
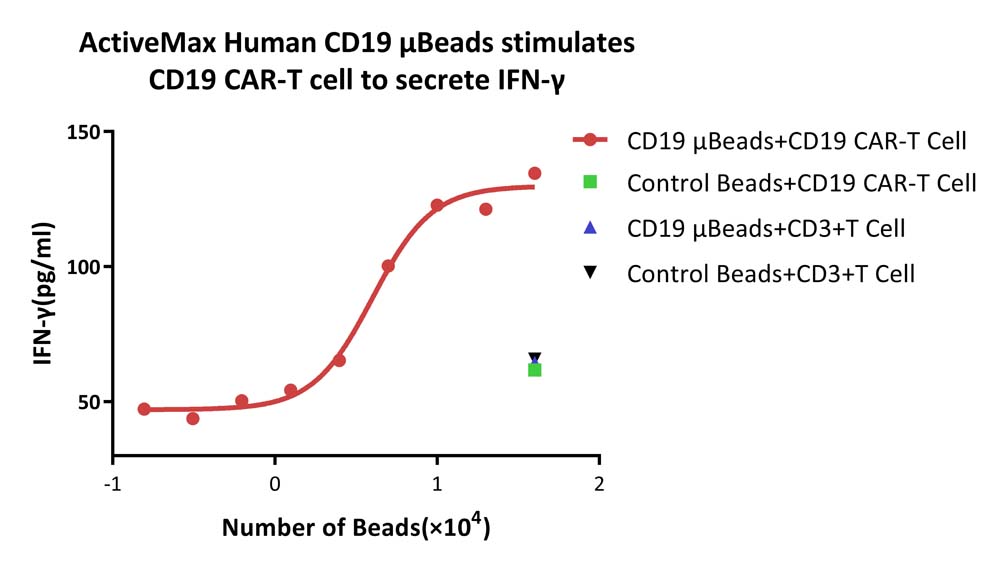
ActiveMax® Human CD19 μBeads, premium grade (for cells) (Cat. No. MBS-C002) can activate CD19-specific CAR-T cells by detecting the secretion of IFN- γ in vitro (Routinely tested).
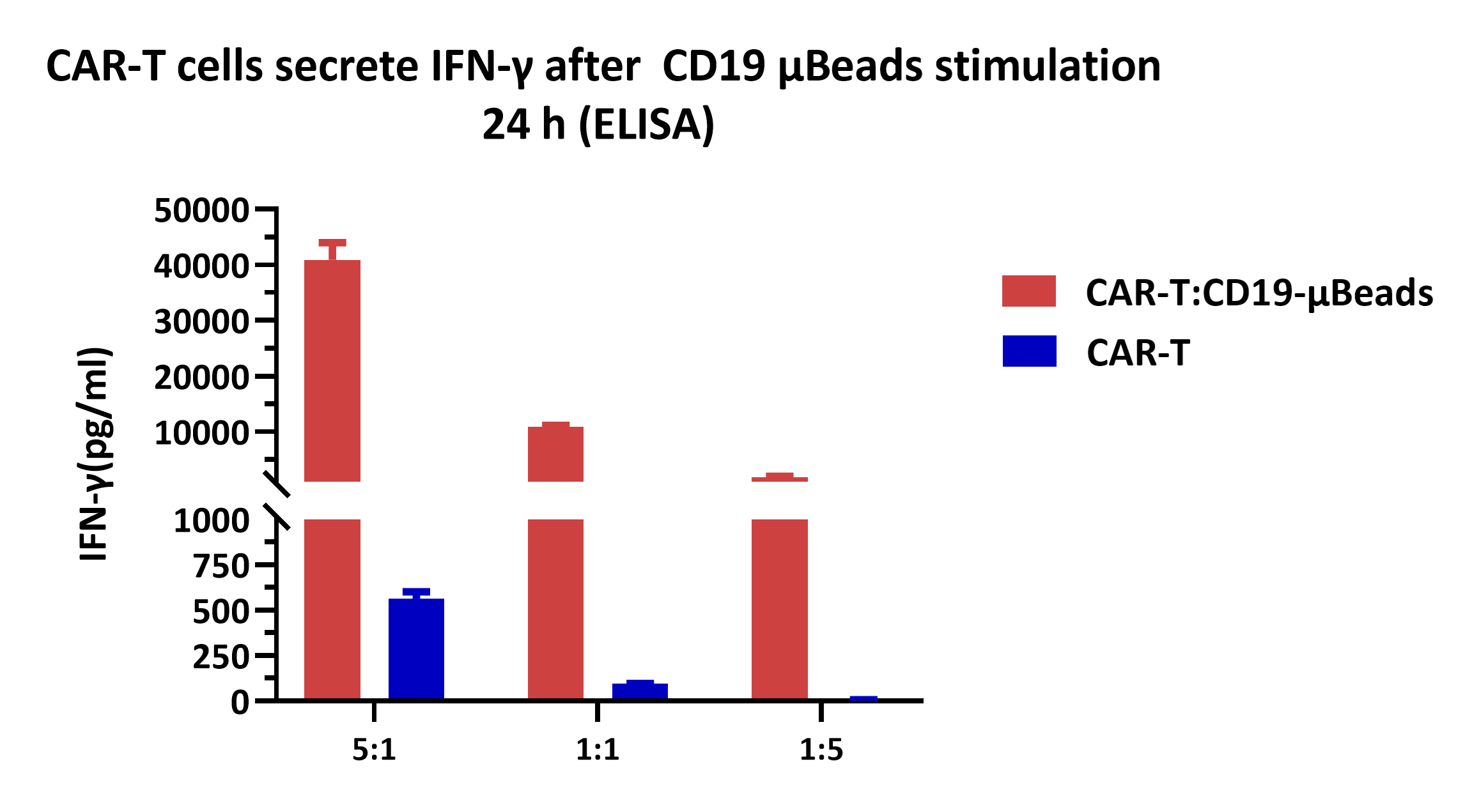
CD19 CAR-T cells were stimulated with ActiveMax® Human CD19 μBeads (MBS-C002) for 24 h , and cell-free supernatants were harvested for evaluating IFN-γ secretion by ELISA. The results showed that CD19 CAR-T cells released significantly larger amounts of IFN-γ into the supernatants in response to CD19 μBeads . Seed the CD19 μBeads in 96-well plates at a density of 2×10⁴ μBeads/well, and co-culture with CD19 CAR-T cells at an effector-to-target (E:T) ratio of 5:1, 1:1, 1:5 in 96-well tissue culture plate, respectively. CD19 CAR-T cells alone were cultured as the control.
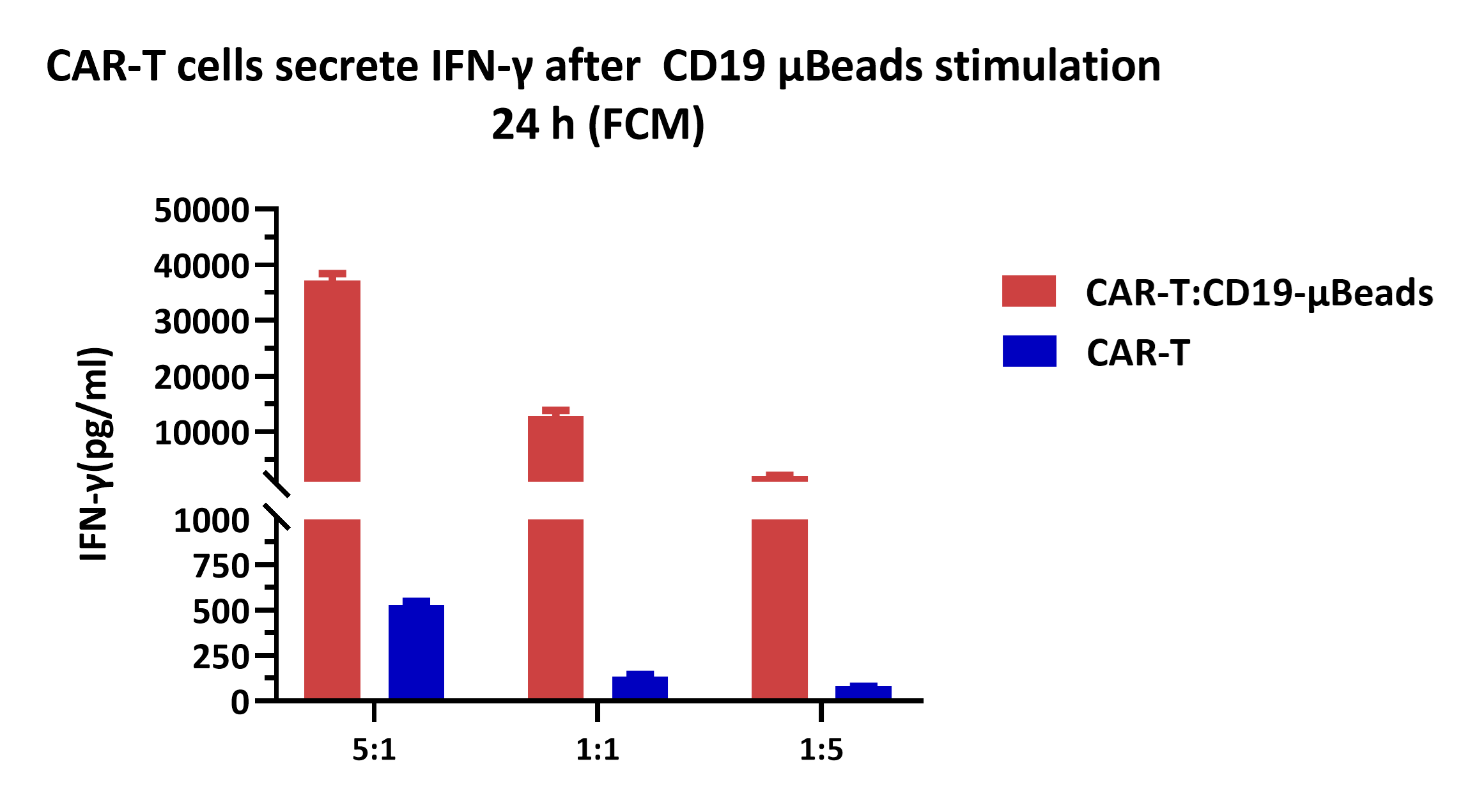
CD19 CAR-T cells were stimulated with ActiveMax® Human CD19 μBeads (MBS-C002) for 24 h , and cell-free supernatants were harvested for evaluating IFN-γ secretion by flow cytomtry. The results showed that CD19 CAR-T cells released significantly larger amounts of IFN-γ into the supernatants in response to CD19 μBeads. Seed the CD19 μBeads in 96-well plates at a density of 2×10⁴ μBeads/well, and co-culture with CD19 CAR-T cells at an effector-to-target (E:T) ratio of 5:1, 1:1, 1:5 in 96-well tissue culture plate, respectively. CD19 CAR-T cells alone were cultured as the control.
应用数据(Application Data)

CD19 CAR-T cells were stimulated with ActiveMax® Human CD19 μBeads (MBS-C002) for 24 h , and cells were harvested for evaluating CD69,CD137 expression by flow cytometry. The results showed that the proportion of CD69 and CD137 in CD19 CAR-T cells was significantly increased after CD19 μBeads stimulation.
Seed the CD19 μBeads in 96-well plates at a density of 2×10⁴ μBeads/well, and co-culture with CD19 CAR-T cells at an effector-to-target (E:T) ratio of 5:1, 1:1, 1:5 in 96-well tissue culture plate, respectively. CD19 CAR-T cells alone were cultured as the control.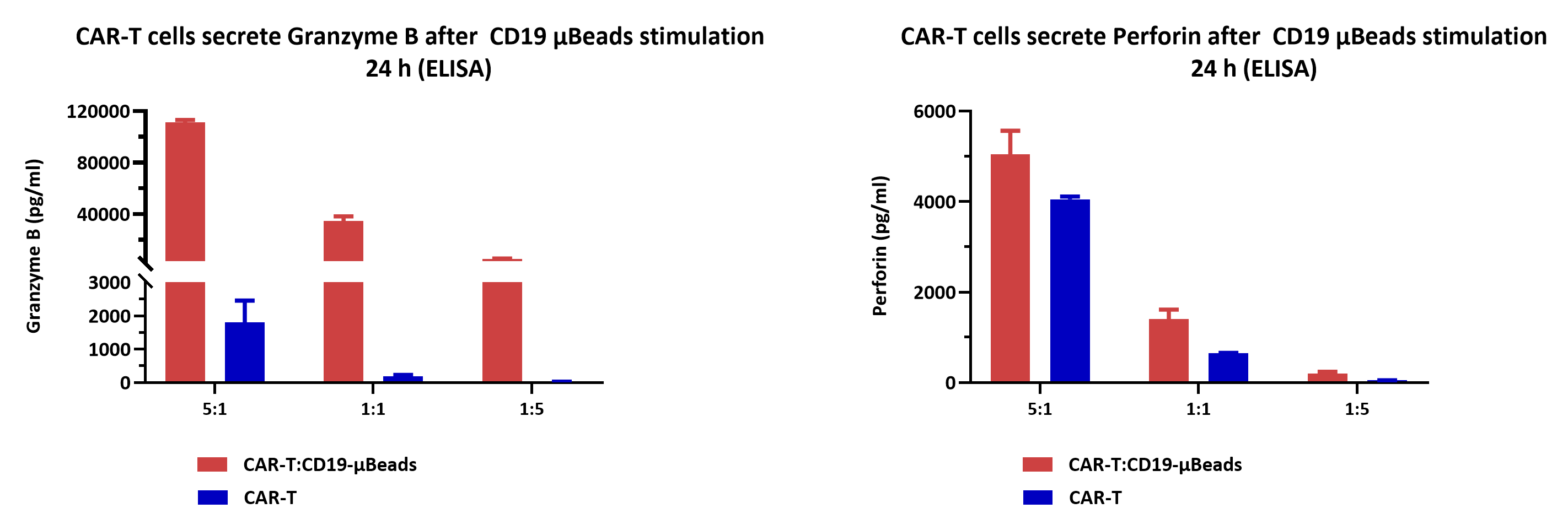
CD19 CAR-T cells were stimulated with ActiveMax® Human CD19 μBeads (MBS-C002) for 24h, and cell-free supernatants were harvested for evaluating Granzyme B, Perforin secretion by ELISA. The results showed that CD19 CAR-T cells released significantly larger amounts of Granzyme B, Perforin into the supernatants in response to CD19 μBeads.
Seed the CD19 μBeads in 96-well plates at a density of 2×10⁴ μBeads/well, and co-culture with CD19 CAR-T cells at an effector-to-target (E:T) ratio of 5:1, 1:1, 1:5 in 96-well tissue culture plate, respectively. CD19 CAR-T cells alone were cultured as the control.
用户评价 发表评论

